 
Multiple methods for analysis of BUN and creatinine have evolved over the years. For the adult female, with her generally lower muscle mass, the normal range is 0.5 to 1.1 mg/dl, or 44 to 97 μmol/L by the enzymatic method. 
For the adult male, the normal range is 0.6 to 1.2 mg/dl, or 53 to 106 μmol/L by the kinetic or enzymatic method, and 0.8 to 1.5 mg/dl, or 70 to 133 μmol/L by the older manual Jaffé reaction. The normal serum creatinine ( sCr) varies with the subject's body muscle mass and with the technique used to measure it. In contrast, the rugged rancher who eats in excess of 125 g protein each day may have a normal BUN of 20 mg/dl. Her higher glomerular filtration rate (GFR), expanded extracellular fluid volume, and anabolism in the developing fetus contribute to her relatively low BUN of 5 to 7 mg/dl. A BUN of 15 mg/dl would represent significantly impaired function for a woman in the thirtieth week of gestation. The range is wide because of normal variations due to protein intake, endogenous protein catabolism, state of hydration, hepatic urea synthesis, and renal urea excretion. The normal range of urea nitrogen in blood or serum is 5 to 20 mg/dl, or 1.8 to 7.1 mmol urea per liter. The BUN, then, is roughly one-half (28/60 or 0.446) of the blood urea. In Europe, the whole urea molecule is assayed, whereas in the United States only the nitrogen component of urea (the blood or serum urea nitrogen, i.e., BUN or SUN) is measured. Both are relatively small molecules (60 and 113 daltons, respectively) that distribute throughout total body water. Creatinine is the product of muscle creatine catabolism. 
Urea is the primary metabolite derived from dietary protein and tissue protein turnover. Urea and creatinine are nitrogenous end products of metabolism. 
0 Comments

 Long Life Coolant Red satisfies or exceeds many major standards and is recommended for major European, Japanese and US car and heavy-duty diesel engines. 
If the product is to be mixed with some heavy duty diesel coolants, the user must continue to add SCA’s (Supplementary Coolant Additives) for heavy-duty service until the next coolant change-out. Long Life Coolant Red is compatible with conventional coolants and can be used for top-up of conventional coolants. 
It is silicate free for improved pump life and contains no environmentally harmful nitrates, borates, phosphates, amines or nitrites. The product is dyed a distinctive red-orange colour for ease of identification. Long Life Coolant Red is an ethylene glycol based antifreeze/coolant concentrate incorporating an advanced formula technology with virtually non-depleting organic acid corrosion inhibitors. Home All Product Categories Coolants & Brake Fluids LONG LIFE COOLANT RED
 Heme proteins include cytochromes, catalase and hemoglobin. Some iron-binding proteins contain heme - a porphyrin ring coordinated with an iron ion. This process is essential to life on Earth, because it’s required for all forms of life for the biosynthesis of nucleotides and amino acids. Iron-dependent enzymes include bacterial nitrogenases, which contain iron-sulfur clusters that catalyze the reduction of nitrogen (N2) to ammonia (NH3) in a process called nitrogen fixation. 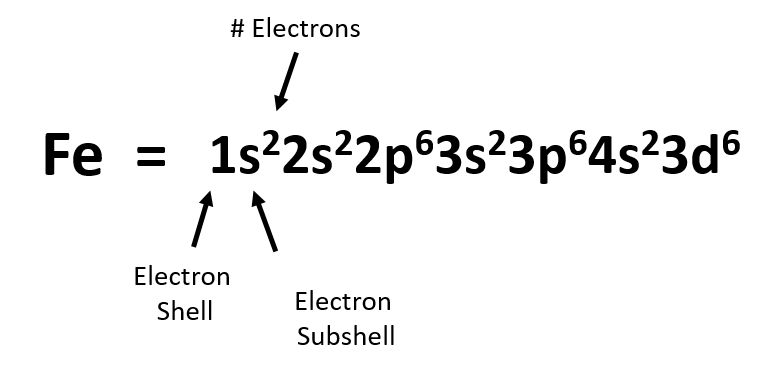
Carrier proteins called ferritins (present in both prokaryotes and eukaryotes) store, transport and safely release iron in areas of need, preventing excess free radicals generated by high-energy iron. Once inside cells, iron associates with carrier proteins and with iron-dependent enzymes. Plants also use sequestration and reduction mechanisms to acquire iron from the rhizosphere, whereas animals obtain iron from dietary sources. Microorganisms can uptake iron from the environment by secreting iron-chelating molecules called siderophores or via membrane-bound proteins that reduce Fe+3 (ferric iron) to a more soluble Fe+2 (ferrous iron) for intracellular transport. Biological systems are exposed constantly to high concentrations of iron in igneous and sedimentary rocks. Iron is crucial to the survival of all living organisms. Iron is present in the Earth’s crust, core and mantle, where it makes up about 35 percent of the planet’s total mass. Each of these supergiant, iron-containing stars only lives for a brief while before violently blasting as a supernova, scattering iron into space and onto rocky planets like Earth. It is produced abundantly in the core of massive stars by the fusion of chromium and helium at extremely high temperatures. Iron occurs naturally in the known universe. Commonly, iron uses two (oxidation state +2) or three (oxidation state +3) of its available electrons to form compounds, although iron oxidation states ranging from -2 to +7 are present in nature. As with other transition metals, a variable number of electrons from iron’s two outermost shells are available to combine with other elements. In February, we have selected iron, the most abundant element on Earth, with chemical symbol Fe (from the Latin word “ferrum”) and atomic number 26.Ī neutral iron atom contains 26 protons and 30 neutrons plus 26 electrons in four different shells around the nucleus. 1 and dissected hydrogen’s role in oxidation-reduction reactions and electrochemical gradients as driving energy force for cellular growth and activity. In iron-deficiency anemia, the heart works harder to pump more oxygen through the body, which often leads to heart failure or disease.We are celebrating the 150th anniversary of Mendeleev’s periodic table by highlighting one or more chemical elements with important biological functions each month in 2019. Each monomer contains a heme group in which an iron ion is bound to oxygen. Hemoglobin is a tetramer that consists of four polypeptide chains.  |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |






 RSS Feed
RSS Feed
